Interesting Case October 2020
Case contributed by:
Roula Katerji MD, Mark Ettel MD
University of Rochester Medical Center
Have an interesting case to share?
Contact HPHS newsletter committee chair Sadhna.Dhingra@ProPath.com
Clinical History
An 86 year old woman presented with generalized abdominal pain, fatigue, and recent jaundice. She has had dysphagia, weight loss, and increased liver enzymes for the past 3 months (most recently AST 106 IU/L, ALT 86 IU/L, alkaline phosphatase 282 IU/L, and total bilirubin 2.3 mg/dL). The patient has a past medical history of hypertension and dilated cardiomyopathy, and has a history of arrhythmia which is now well-controlled with a pacemaker and an amiodarone dose of 200 mg tablets twice daily. Her BMI is 30.2 kg/m2, she is a former smoker, occasionally consumes alcohol, and denies illicit drug abuse.
Her hepatitis viral serology results were negative. Her ANA was mildly elevated (1:80) but autoimmune serology results were otherwise negative.
A liver biopsy was performed.
Pathologic Findings
Liver core biopsies showed numerous ballooned hepatocytes (Figure 1), extensive Mallory bodies (Figure 2), mild steatosis, and numerous foci of neutrophilic satellitosis (Figure 3). Trichrome stain (Figure 4) highlighted dense pericellular and periportal fibrosis indicating chronicity.
Figure 1
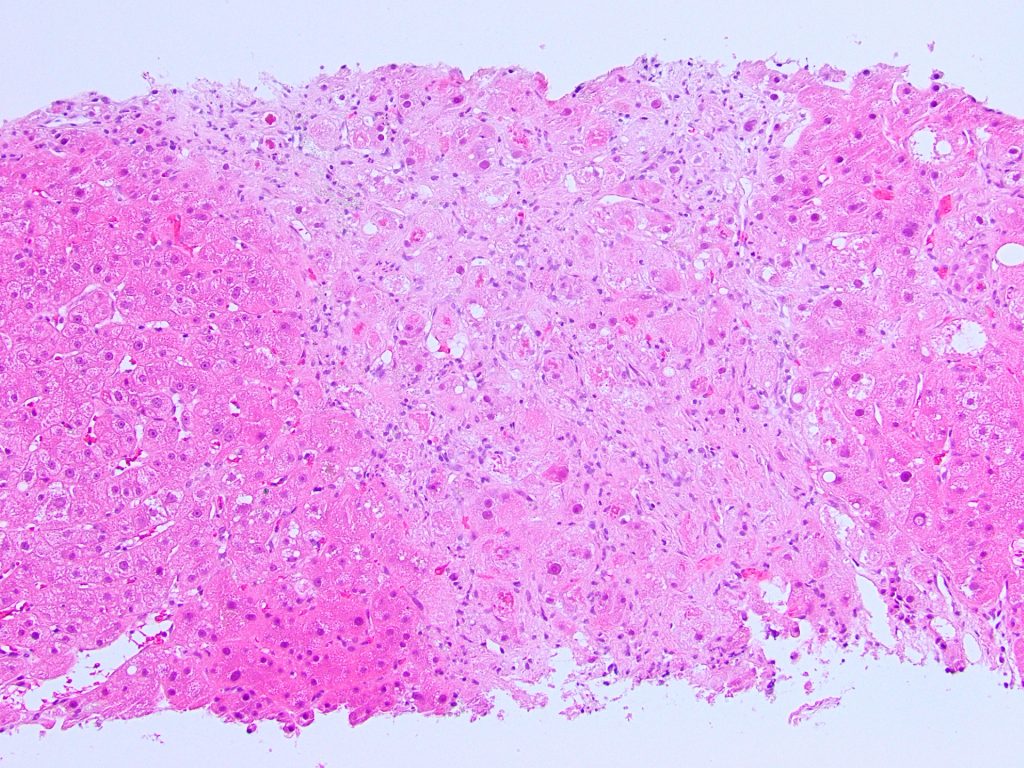
Figure 2
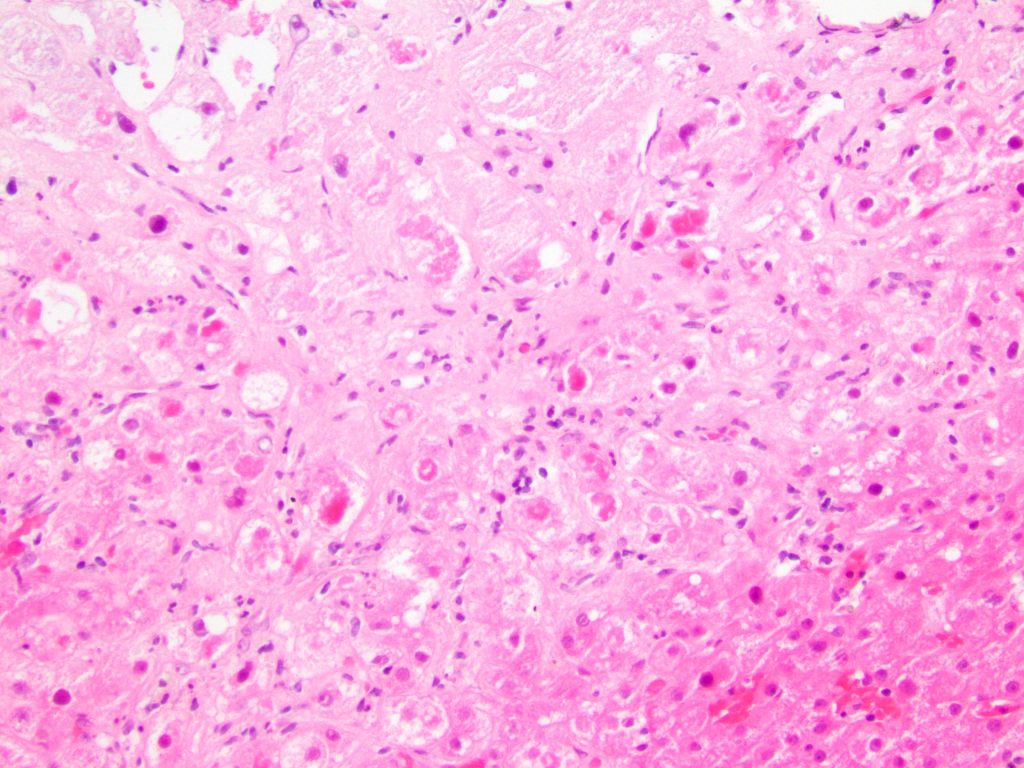
Figure 3
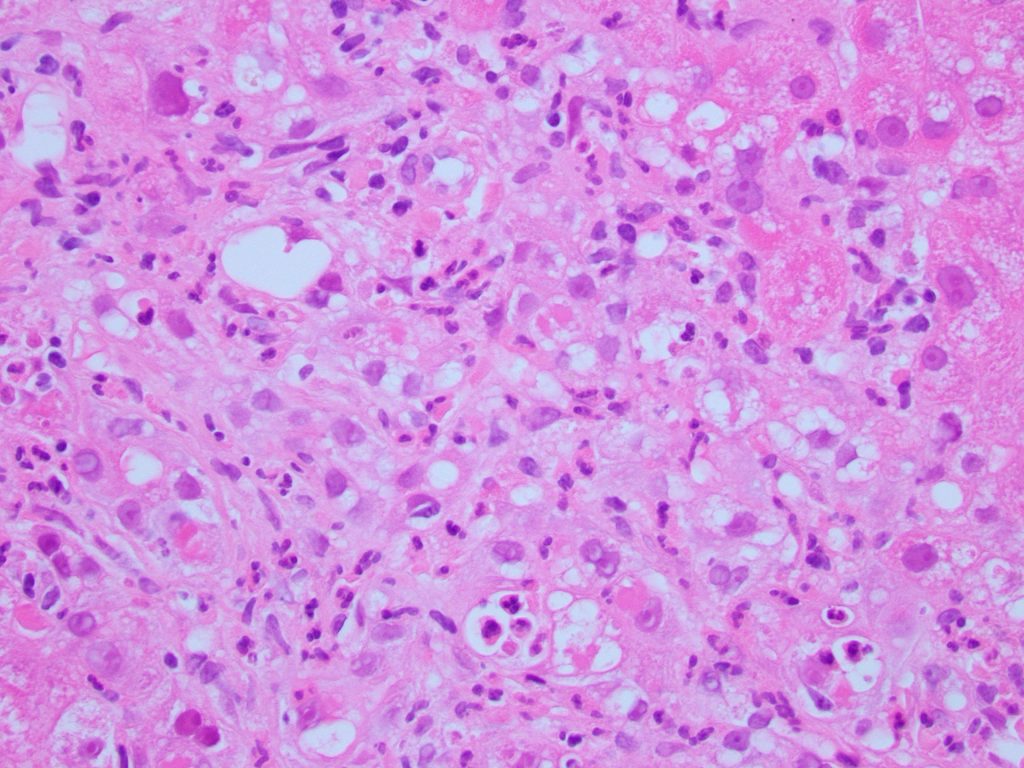
Figure 4
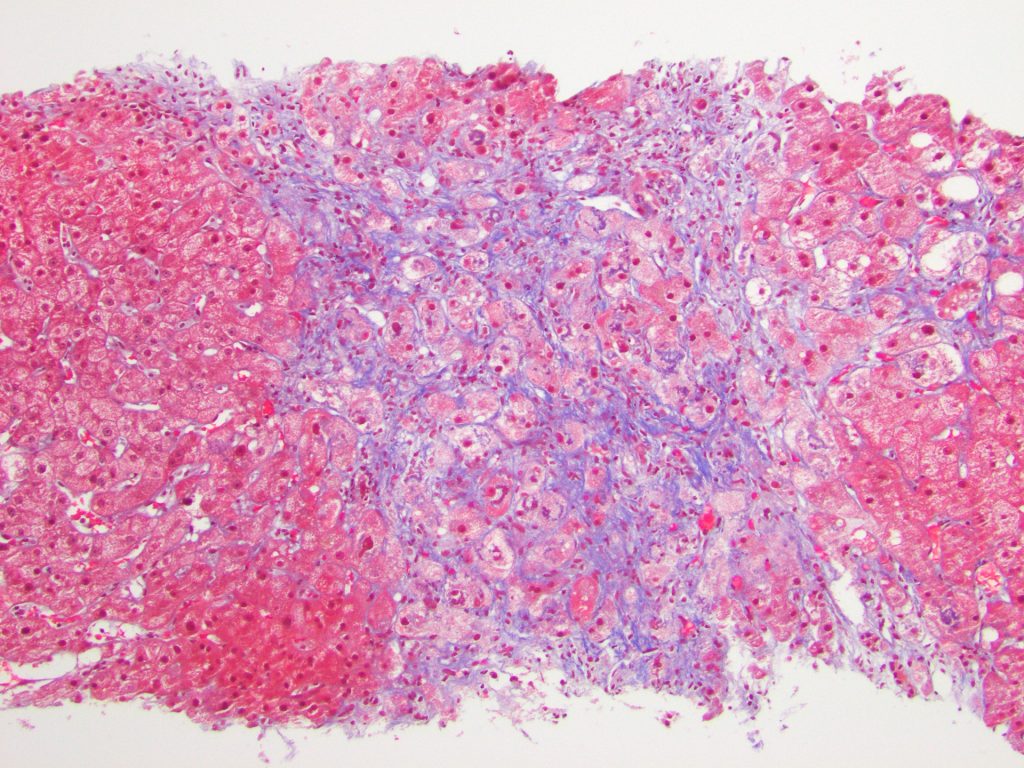
Diagnosis
The histologic findings and the patient’s clinical presentation were most consistent with amiodarone induced acute liver toxicity.
Discussion
Amiodarone is an antiarrhythmic medication used to treat supraventricular and ventricular arrhythmias. It has a broad spectrum of side effects on different organs including the liver.
The liver injury might happen at any time during taking the medication and can be asymptomatic for months or even years before clinical presentation. Amiodarone causes liver damage by mechanisms likely including but not limited to phospholipidosis. Amiodarone causes phospholipidosis by accumulation of the drug and its metabolites (N-desmethylamiodarone) in lysosomes inhibiting phospholipidases A1 and A2, and by directly binding to lysosomes to form indigestible complexes [1, 2]. However, some patients on amiodarone develop phospholipidosis without further evidence of liver injury, so the mechanism of liver injury may be more complex [3].
On histologic examination biopsy findings show changes similar to severe alcoholic liver injury [4], macrovesicular and microvesicular steatosis are seen alongside extensive hepatocyte ballooning, necrosis, neutrophilic satellitosis, and numerous Mallory-Denk bodies (MDB). Although these features including hepatocyte ballooning and MDB are not specific to amiodarone toxicity, they are rarely as prominent when occurring secondary to other etiologies such as alcoholic or nonalcoholic steatohepatitis [5].
Although amiodarone-induced liver injury can be striking for the aforementioned reasons, ultimately it is a diagnosis of exclusion, and extensive workup is usually done to exclude other causes of steatohepatitis. Hence the striking appearance of a severe steatohepatitis as seen in our case should prompt a review of the medication list, especially if the clinical history makes other risk factors appear unlikely as in our patient who only occasionally drinks alcohol and is thus unlikely to have a severe alcoholic steatohepatitis.
Upon diagnosis of amiodarone-induced liver injury, the treatment of choice is drug discontinuation, although levels of amiodarone can persist in tissue for weeks following withdrawal, and drug cessation does not guarantee reversal of injury.
Learning points
1. Amiodarone-induced hepatic toxicity can be asymptomatic for a long time prior to clinical presentation, but due to the chronic course it can be more likely to present with fibrosis than other forms of drug-induced liver injury.
2. Extensive Mallory hyaline and lysosomal inclusions can be a strong indication for amiodarone-induced hepatic toxicity.
3. Because amiodarone-induced hepatic toxicity has a close pathological resemblance to alcoholic steatohepatitis, review of clinical history is essential.
References
1. Hostetler, K.Y., et al., Role of phospholipase A inhibition in amiodarone pulmonary toxicity in rats. Biochimica et Biophysica Acta (BBA) – Lipids and Lipid Metabolism, 1986. 875(2): p. 400-405.
2. Pirovino, M., et al., Amiodarone-induced hepatic phospholipidosis: Correlation of morphological and biochemical findings in an animal model. Hepatology, 1988. 8(3): p. 591-598.
3. Hussain, N., A. Bhattacharyya, and S. Prueksaritanond, Amiodarone-Induced Cirrhosis of Liver: What Predicts Mortality? ISRN Cardiology, 2013. 2013: p. 617943.
4. Lewis, J.H., et al., Histopathologic analysis of suspected amiodarone hepatotoxicity. Hum Pathol, 1990. 21(1): p. 59-67.
5. Basaranoglu, M., et al., Mallory-Denk Bodies in chronic hepatitis. World J Gastroenterol, 2011. 17(17): p. 2172-7.
