Interesting Case: Jan 2018
CLINICAL HISTORY:
A 70- year-old man presented with right upper quadrant pain for 6 months, fatigue, and weight loss of 50 pounds over the past 2 years. Liver function tests were abnormal. AFP was 25,000 ug/mL. The patient did not have known liver disease, or history of malignancy.
IMAGING FINDINGS (COMPUTED TOMOGRAPHY):
There was a heterogenous mass in the right hepatic lobe measuring 23.3 x 12.3 x 13.8 cm. The background liver demonstrated a nodular contour concerning for underlying cirrhosis. The intrahepatic portion of the right portal vein was not visualized. There was a low density extending to the left portal vein concerning for involvement/invasion of the portal vein.
LIVER BIOPSY:
Figure 1: H&E (10X)
Figure 2: H&E (100X)
Figure 3: H&E (100X)
Figure 4: H&E (100X)
LIVER BIOPSY FINDINGS:
The tumor demonstrates heterogeneous growth pattern.
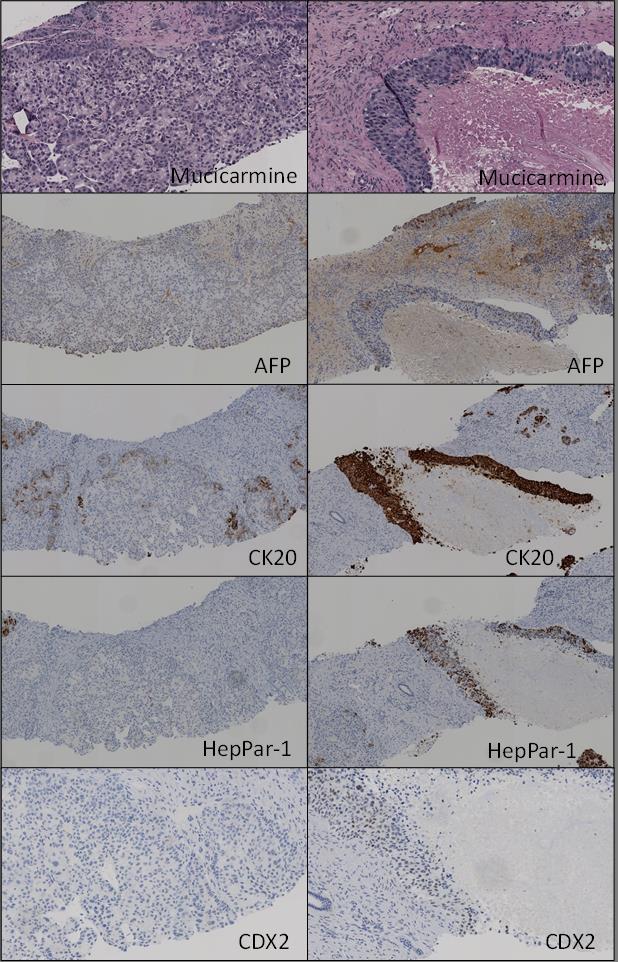
One area shows a poorly differentiated carcinoma characterized by solid nests of tumor cells with frequent central necrosis. In some areas, small acinar/glandular structures bud off the larger nests. Immunohistochemical stains show that this component is positive for HepPar-1 (patchy), AFP (patchy), CK20 (diffuse and strong) and CDX2 (diffuse and weak) and is negative for CK7.
Another area shows conventional hepatocellular carcinoma (HCC) with trabecular/pseudoacinar growth pattern and patchy clearing of the tumor cell cytoplasm. Immunohistochemical stain for CK20 shows occasional positive tumor cells at the periphery of the HCC nests while immunohistochemical stain for CK7 stains native bile ducts and and shows weak cytoplasmic staining of occasional tumor cells at the periphery. This component is negative for HepPar-1, AFP and CDX2.
Immunohistochemical stain for Ki67 shows that the proliferation index is higher in the poorly differentiated carcinoma component.
In both components mucicarmine is negative for mucin, while immunohistochemical stain for glypican-3 shows focal equivocal staining. Immunohistochemical stain for CD10 does not show canalicular pattern staining and synaptophysin, CA19-9, MART1, NapsinA, PSA, and S-100 are negative in both components.
DIAGNOSIS:
HEPATOCELLULAR CARCINOMA, POORLY DIFFERENTIATED.
DISCUSSION:
Nuclear CDX2 staining in a carcinoma usually indicates that the tumor originated from the gastrointestinal (GI) tract [1]. When combined with CK20 positivity, colonic origin is suspeted. Immunoreactivity for CDX2 and CK20 is rare in conventional HCC. In our case, the conventional HCC component was negative for CDX2 and CK20. (Figure 2, Figure 6 left column). On the other hand, the poorly differentiated carcinoma component exhibited a weak but diffuse CDX2 expression and strong CK20 positivity (Figure 4, Figure 6 right column).
In a recent study, Shah et al, for the first time, demonstrated that a small subset (9 of 172 cases; 5%) of HCCs can be CDX2 positive. Interestingly, more than half of this small subset (5 of 9; 56%) was poorly differentiated. Moreover, 5 of 16 (31%) poorly differentiated HCC showed nuclear CDX2 expression whereas 4 of 156 (3%) moderately differentiated HCC were CDX2 positive. In their study, arginase 1 was also positive in all 9 CDX2 positive tumors, glypican 3 was positive in 5 of 5 tumors, and CK20 was negative in 6 of 6 cases, when performed. [2]
CK20 positivity in HCC is rare, but has also been reported. Mourra et al. reported a case of moderately differentiated HCC with diffuse and strong CK20 positivity in the setting of concurrent rectal adenocarcinoma. The HCC was positive for glypican-3 and HepPar-1 while the rectal adenocarcinoma was negative for both. The authors reviewed the literature and reported that the incidence of CK20 reactivity in HCCs is variable, ranging from 0% to 14% without any correlation with histologic grade of HCC. [3]
In order to determine an optimal panel of immunomarkers to diagnose HCC, Nguyen et al employed 5 immunostains – HepPar-1, polyclonal CEA (pCEA), glypican-3, arginase 1 and bile salt export pump transporter (BSEP) – on 79 HCCs, and concluded that arginase 1 is the most sensitive immunomarker in detecting HCC at all levels of differentiation, when the “positivity” was defined as >50% of tumor cells being stained. The combination of arginase 1 and glypican-3 had the highest sensitivity (100%) for diagnosing poorly differentiated HCC. [4]
Our case highlights that co-expression of CDX2 and CK20 does occur in poorly differentiated HCC. While scattered acinar/glandular structure in the periphery of the poorly differentiated component raises the possibility of mixed HCC-cholangiocarcinoma (Figure 3), this is not favored given its negativity for mucicarmine and CK7.
The diagnosis of HCC in our case was relatively straightforward given the clinical presentation including the absence of other gastrointestinal tract tumor. However, this is an unusual and educational case from a pathologic standpoint. The co-expression of CDX2 and CK20 in HCC would pose a diagnostic challenge especially when the biopsy material is limited or if there was compounding clinical history, such as concurrent colon cancer.
Learning point: This case demonstrates aberrant expression of CK20 and CDX2 in poorly differentiated HCC. Marked central necrosis in conjunction with the immunoprofile raised the possibility of metastatic carcinoma from the gastrointestinal tract. However, adjacent conventional HCC-like area (Figure2), patchy HepPar-1 positivity and clinical presentation (single mass in a background of cirrhosis with very high AFP) support that the poorly differentiated component with aberrant immunoprofile also represents HCC. Co-expression of CK20 and CDX2 may be seen in HCC, thus this immunoprofile does not exclude a diagnosis of HCC, especially in a poorly differentiated tumor. The addition of arginase1 may be helpful in similar cases since this appears to be the most sensitive immunomarker for HCC of all differentiation, and a combined use of arginase 1 and glypican-3 showed the highest sensitivity for poorly differentiated hepatocellular carcinoma. [4]
REFERENCES:
1) Werling RW, Yaziji H, Bacchi CE, Gown AM. CDX2, a highly sensitive and specific marker of adenocarcinomas of intestinal origin: an immunohistochemical survey of 476 primary and metastatic carcinomas. Am J Surg Pathol 2003;27:303-310.
2) Shah SS, Wu TT, Torbenson MS, Chandan VS. Aberrant CDX2 expression in Hepatocellular Carcinomas: An Important Diagnostic Pitfall. Hum Pathol. 2017;64: 13-18. doi: 10.1016/j.humpath.2016.12.029.
3) Mourra N, Azizi L. CK20 positivity in hepatocellular carcinoma: a potential diagnostic pitfall in liver biopsy. Appl Immunohistochem Mol Morphol. 2013;21(1):94-95. doi: 10.1097/PAI.0b013e31824c4c4a.
4) Nguyen T, Phillips D, Jain D, et al. Comparison of 5 Immunohistochemical Markers of Hepatocellular Differentiation for the Diagnosis of Hepatocellular Carcinoma. Arch Pathol Lab Med. 2015;139(8):1028-34. doi: 10.5858/arpa.2014-0479-OA.
Case submitted by
Tony El-Jabbour, MD; Resident, Pathology and Laboratory Medicine, Albany Medical College, Albany, NY.
Hwajeong Lee, MD; Associate Professor, Pathology and Laboratory Medicine, Albany Medical College, Albany, NY.