Interesting Case August 2016
Clinical History
A 43 year old woman with a previous history of ductal breast carcinoma, status post chemo -radiation and surgical therapy, was found to have a liver lesion detected on surveillance imaging.
Imaging Findings
Abdominal MRI demonstrated a 3.1 cm lesion in the right lobe of the liver. The lesion showed prompt arterial enhancement and gradual washout. Fine needle aspiration (FNA) and needle core biopsy was performed. FNA disproved metastatic disease and prompted a partial hepatectomy.
Photomicrographs
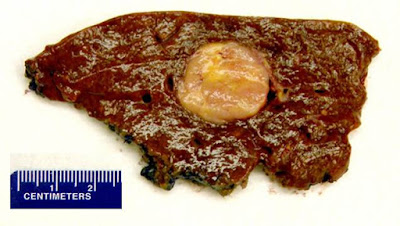 |
| Figure 1. Gross photograph of the right hepatectomy specimen |
 |
| Figure 2. H&E (400X) |
 |
| Figure 3. TFE-3 immunohistochemistry (400X) |
 |
| Figure 4. HMB45 immunohistochemistry (400X) |
Liver Biopsy Findings
The liver biopsy showed an infiltrative neoplastic process with a solid to vaguely organoid growth pattern. The tumor was composed of large plump cells showing an epithelioid and plasmacytoid appearance. The tumor cells had abundant eosinophilic cytoplasm with low nuclear to cytoplasmic ratios and frequent multinucleated giant cells. Mitotic figures were rare. Necrosis was not detected. The background liver exhibited no features of chronic hepatitis or cirrhosis.
Immunohistochemistry showed that the tumor cells were reactive with antibodies to TFE-3 (Fig 3), HMB-45 (Fig 4), cathepsin K, and MITF. The tumor cells were negative for AE1/3, CAM 5.2, ER, PR, S100, Melan A, arginase 1, INI-1, CD31, E-cadherin, SMA, desmin, and inhibin. The electron microscopy study failed to reveal intracytoplasmic crystals. The FISH assay for rearrangement of TFE-3 locus gene at Xp11.2 was negative.
Diagnosis
Perivascular epithelioid cell neoplasm (“PEComa”)
Major Learning Points
1. The presence of an organoid growth pattern and epithelioid or plasmacytoid cells should raise the possibility of PEComa.
2. PEComa is a unique tumor composed of histologically and immunohistochemically distinct perivascular epithelioid cells with coexpression of both melanocytic and smooth muscle markers.
3. The presence of HMB-45-positive myoid cells is diagnostic.
Discussion
Perivascular epithelioid cell neoplasm (PEComa) belongs to a family of tumors composed of histologically and immunohistochemically distinctive perivascular epithelioid cells. This group includes renal and extra-renal angiomyolipoma, lymphangiomatosis, clear cell “sugar” tumor of the lung, myelomelanocytic tumor, and abdominopelvic sarcoma of perivascular epithelioid cells. PEComa of the liver is exceedingly rare. PEComas have been predominantly described in women with the average age of 46. Since PEComa is associated with tuberous sclerosis (6-10%), overactive mTOR pathway is possibly contributing to tumorigenesis.
Grossly the tumor occurs as a yellow to gray mass, which may reach up to 20 cm in size. Microscopically, PEComas are composed of clear to slightly eosinophilic cells that grow in distinctive nested or sometimes sheet-like patterns. The cells organize in a radial fashion around blood vessels. The blood vessel types range from thin spider web-like capillary networks to dilated, hyalinized, thick-walled arterioles. Both epithelioid and myoid elements are prominent, with the epithelioid cells typically occupying the more immediate perivascular space and the spindle-shaped myoid cells farther away. Multinucleated giant cells are characteristic. Many cells may demonstrate cytoplasmic distention with glycogen or lipid. PEComas are generally considered benign neoplasms, if they lack malignant morphologic features. Tumors that are “symplastic” or large in size are considered uncertain in terms of malignant potential. The dimorphic cellular composition of this tumor makes fine needle aspiration and core biopsy diagnosis difficult as it was witnessed in our case. Higher grade and cellularity correspond to higher malignant potential. If more than two “worrisome” histologic features are identified, the tumor is considered malignant.
The immunohistochemical features to distinguish PEComas from other neoplasms is the unique coexpression of both melanocytic and smooth muscle markers. The most important diagnostic criterion, according to WHO classification of tumors, is the presence of HMB-45-positive myoid cells. PEComas with a predominant component of clear cells also show extensive TFE-3 and cathepsin K positivity, but they are not typically associated with tuberous sclerosis complex mutations.
The chief histologic differential diagnosis in our case included metastatic mammary carcinoma, alveolar soft part sarcoma and malignant melanoma. Review of the previous mastectomy, the negative immunohistochemistry for breast markers and the unusual morphology largely ruled out a metastatic breast carcinoma. The organoid growth pattern with loosely cohesive epithelioid cells raised the possibility of alveolar soft part sarcoma. This, however, was excluded by electron microscopy and the lack of TFE-3 gene Xp11 rearrangement. Lack of malignant features such as frequent mitotic figures and necrosis helped excluded metastatic malignant melanoma. Currently, the only curative option for liver PEComa is surgical excision with wide margins. Adjuvant therapy has no defined role in the treatment of metastatic disease. Rapamycin, an inhibitor of mTOR pathway, may be beneficial for patients with metastatic disease.
References
Enzinger & Weiss’s. Soft tissue Tumors. Ch. 36. Perivascular epithelioid cell family of tumors. 5th ed. 2008. Mosby Inc, Elsevier. Philadelphia.
Colden C, Walsh NJ, Kruse EJ: Perivascular Epithelioid Cell Tumor of the Liver. The American surgeon 2015, 81:e345-6.
Tsui WM, Colombari R, Portmann BC, Bonetti F, Thung SN, Ferrell LD, Nakanuma Y, Snover DC, Bioulac-Sage P, Dhillon AP: Hepatic angiomyolipoma: a clinicopathologic study of 30 cases and delineation of unusual morphologic variants. The American journal of surgical pathology 1999, 23:34-48
Agaram NP, Sung YS, Zhang L, Chen CL, Chen HW, Singer S, Dickson MA, Berger MF, Antonescu CR: Dichotomy of Genetic Abnormalities in PEComas With Therapeutic Implications. The American journal of surgical pathology 2015, 39:813-25.
Kenerson H, Folpe AL, Takayama TK, Yeung RS: Activation of the mTOR pathway in sporadic angiomyolipomas and other perivascular epithelioid cell neoplasms. Human pathology 2007, 38:1361-71.
This case was submitted by:
Natalia I. Rush, MD, Resident, Department of Pathology and Laboratory Medicine, Indiana University School of Medicine, Indianapolis, IN
Jingmei Lin, MD PhD, Assistant Professor, Department of Pathology and Laboratory Medicine, Indiana University School of Medicine, Indianapolis, IN
